draw the lewis structure for so42-. how many equivalent resonance structures can be drawn?
Resonance
- Folio ID
- 2006
Resonance is a mental exercise within the Valence Bail Theory of bonding that describes the delocalization of electrons inside molecules. It involves constructing multiple Lewis structures that, when combined, correspond the full electronic structure of the molecule. Resonance structures are used when a unmarried Lewis structure cannot fully depict the bonding; the combination of possible resonance structures is defined as a resonance hybrid, which represents the overall delocalization of electrons within the molecule. In general, molecules with multiple resonance structures will be more stable than one with fewer and some resonance structures contribute more to the stability of the molecule than others - formal charges aid in determining this.
Introduction
Resonance is a fashion of describing delocalized electrons inside sure molecules or polyatomic ions where the bonding cannot be expressed by a single Lewis formula. A molecule or ion with such delocalized electrons is represented by several resonance structures. The nuclear skeleton of the Lewis Construction of these resonance structures remains the aforementioned, simply the electron locations differ. Such is the case for ozone (\(\ce{O3}\)), an allotrope of oxygen with a 5-shaped construction and an O–O–O angle of 117.5°. Allow's motivate the word past edifice the Lewis construction for ozone.
1. Nosotros know that ozone has a V-shaped structure, and then one O atom is central:

two. Each O atom has 6 valence electrons, for a full of xviii valence electrons.
iii. Assigning ane bonding pair of electrons to each oxygen–oxygen bond gives
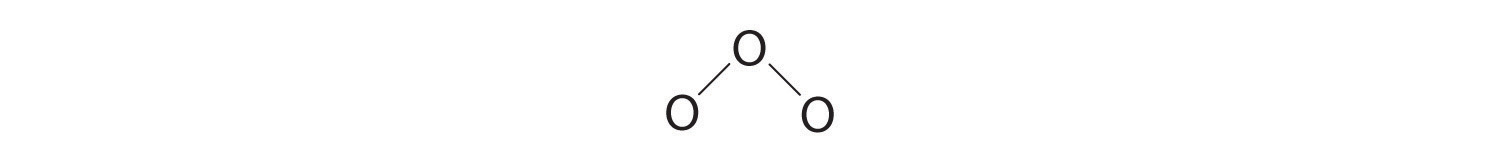
with fourteen electrons left over.
4. If we place iii lonely pairs of electrons on each terminal oxygen, we obtain

and have 2 electrons left over.
5. At this signal, both last oxygen atoms take octets of electrons. We therefore place the final ii electrons on the central atom:
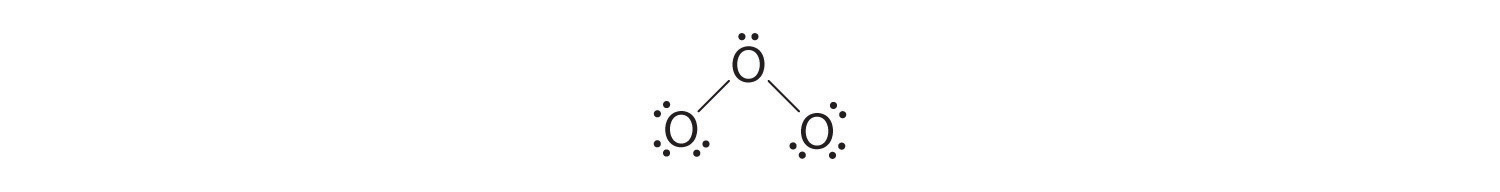
half-dozen. The primal oxygen has only 6 electrons. Nosotros must convert one lone pair on a terminal oxygen atom to a bonding pair of electrons—but which ane? Depending on which one we choose, we obtain either
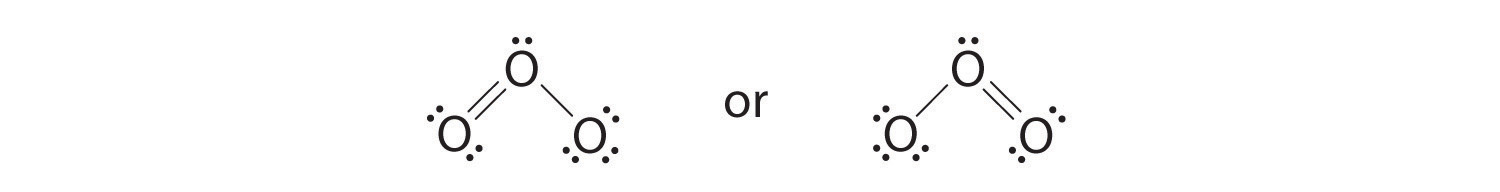
Which is correct? In fact, neither is correct. Both predict one O–O unmarried bond and one O=O double bail. As you will learn, if the bonds were of different types (one unmarried and ane double, for example), they would have different lengths. It turns out, all the same, that both O–O bond distances are identical, 127.2 pm, which is shorter than a typical O–O single bond (148 pm) and longer than the O=O double bail in O2 (120.7 pm).
Equivalent Lewis dot structures, such as those of ozone, are called resonance structures. The position of the atoms is the same in the diverse resonance structures of a compound, but the position of the electrons is different. Double-headed arrows link the different resonance structures of a compound:
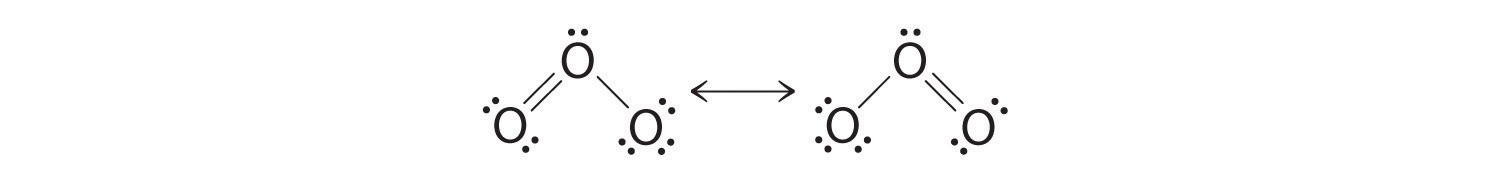
The double-headed pointer indicates that the bodily electronic structure is an boilerplate of those shown, not that the molecule oscillates between the two structures.
When it is possible to write more one equivalent resonance structure for a molecule or ion, the actual structure is the average of the resonance structures.
The electrons appear to "shift" between different resonance structures and while non strictly correct equally each resonance structure is just a limitation of using the Lewis structure perspective to describe these molecules. A more than authentic clarification of the electron structure of the molecule requires considering multiple resonance structures simultaneously.
Delocalization and Resonance Structures Rules
- Resonance structures should have the same number of electrons, do not add or decrease whatsoever electrons. (check the number of electrons past simply counting them).
- Each resonance structures follows the rules of writing Lewis Structures.
- The hybridization of the structure must stay the same.
- The skeleton of the structure can non exist inverse (simply the electrons motion).
- Resonance structures must also have the aforementioned number of lonely pairs.
"Choice the Correct Arrow for the Task"
Most arrows in chemical science cannot exist used interchangeably and care must be given to selecting the correct arrow for the chore.
- \(\leftrightarrow\): A double headed arrow on both ends of the arrow between Lewis structures is used to bear witness resonance
- \(\rightleftharpoons\): Double harpoons are used to designate equilibria
- \(\rightharpoonup\): A unmarried harpoon on one end indicates the movement of i electron
- \( \rightarrow \): A double headed pointer on i finish is used to signal the movement of two electrons
Example \(\PageIndex{2}\): Carbonate Ion
Identify the resonance structures for the carbonate ion: \(\ce{CO3^{2-}}\).
Solution
1. Because carbon is the least electronegative element, nosotros place it in the cardinal position:

2. Carbon has 4 valence electrons, each oxygen has vi valence electrons, and in that location are two more than for the −2 charge. This gives 4 + (three × 6) + 2 = 24 valence electrons.
3. Six electrons are used to form three bonding pairs between the oxygen atoms and the carbon:
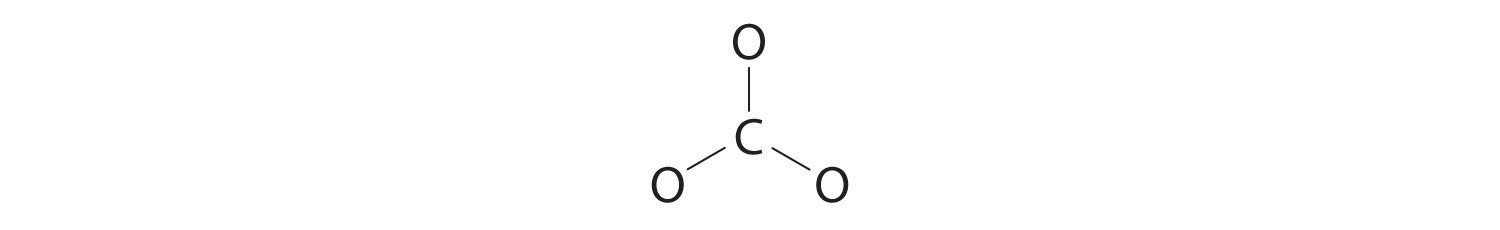
four. We split up the remaining 18 electrons equally amongst the iii oxygen atoms by placing three lone pairs on each and indicating the −2 charge:
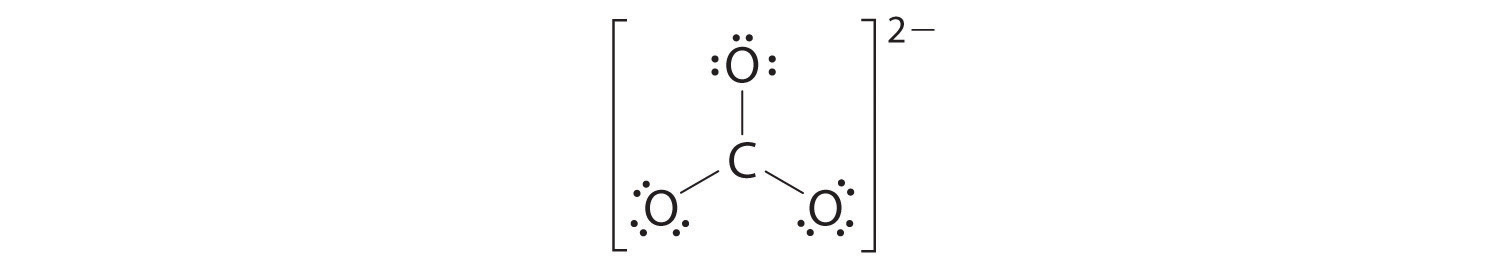
five. No electrons are left for the central atom.
half dozen. At this point, the carbon cantlet has only half-dozen valence electrons, and then we must accept one lone pair from an oxygen and use it to course a carbon–oxygen double bond. In this example, however, in that location are three possible choices:
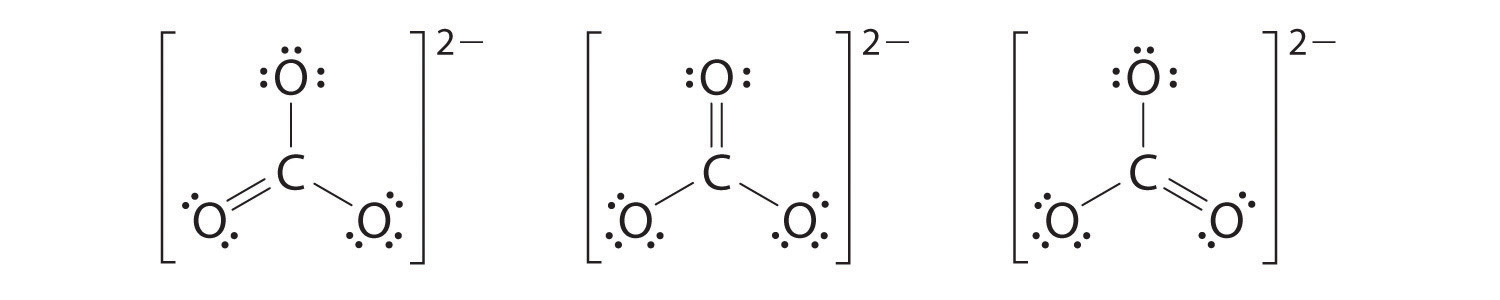
As with ozone, none of these structures describes the bonding exactly. Each predicts one carbon–oxygen double bond and two carbon–oxygen single bonds, but experimentally all C–O bond lengths are identical. We tin can write resonance structures (in this case, three of them) for the carbonate ion:
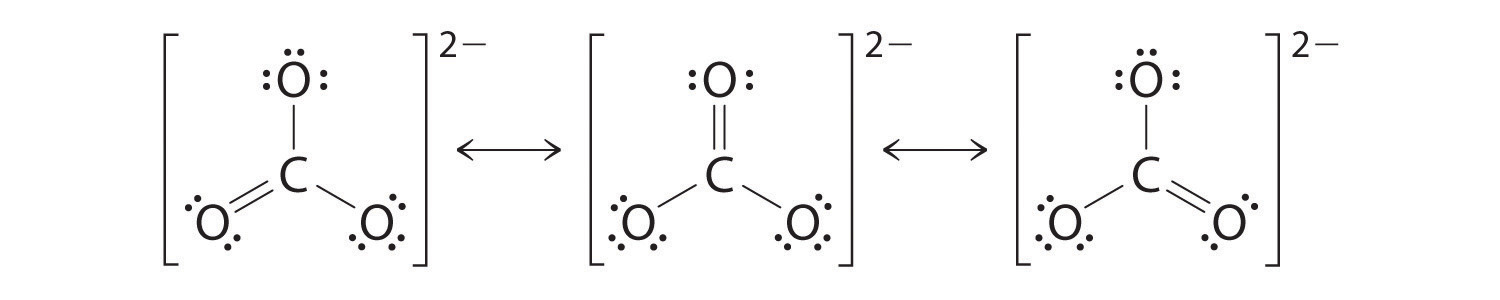
The actual construction is an average of these three resonance structures.
Like ozone, the electronic structure of the carbonate ion cannot be described by a single Lewis electron structure. Different O3, though, the actual structure of CO3 2− is an average of iii resonance structures.
Using Formal Charges to Place viable Resonance Structures
While each resonance structure contributes to the total electronic structure of the molecule, they may non contribute equally. Assigning Formal charges to atoms in the molecules is one mechanism to identify the viability of a resonance structure and determine its relative magnitude among other structures. The formal charge on an cantlet in a covalent species is the internet accuse the atom would conduct if the electrons in all the bonds to the atom were every bit shared. Alternatively the formal accuse on an cantlet in a covalent species is the net charge the atom would bear if all bonds to the atom were nonpolar covalent bonds. To decide the formal charge on a given atom in a covalent species, use the following formula:
\[\text{Formal Accuse} = (\text{number of valence electrons in free orbital}) - (\text{number of lone-pair electrons}) - \frac{one}{2} (\text{ number bail pair electrons}) \label{FC}\]
Rules for estimating stability of resonance structures
- The greater the number of covalent bonds, the greater the stability since more atoms volition have consummate octets
- The structure with the to the lowest degree number of formal charges is more stable
- The construction with the least separation of formal charge is more than stable
- A structure with a negative charge on the more electronegative atom will exist more stable
- Positive charges on the least electronegative atom (most electropositive) is more stable
- Resonance forms that are equivalent have no difference in stability and contribute every bit (eg. benzene)
Instance \(\PageIndex{3}\): Thiocyanate Ion
Consider the thiocyanate (\(CNS^-\)) ion.
Solution
1. Discover the Lewis Construction of the molecule. (Call back the Lewis Construction rules.)
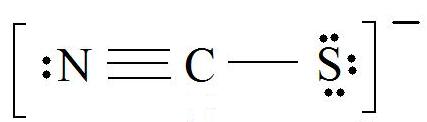
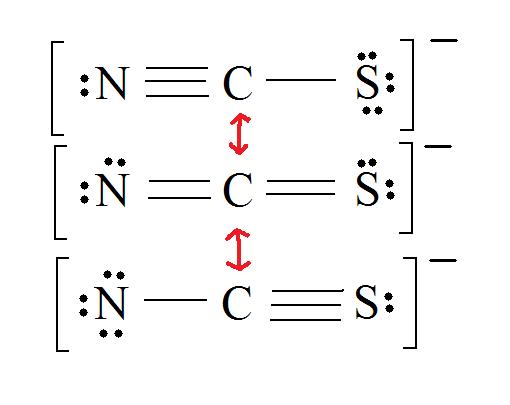
2. Resonance: All elements want an octet, and we tin can do that in multiple ways past moving the terminal atom's electrons around (bonds too).
3. Assign Formal Charges via Equation \ref{FC}.
Formal Accuse = (number of valence electrons in free orbital) - (number of lonely-pair electrons) - ( \( \frac{i}{2} \) number bail pair electrons)
Remember to determine the number of valence electron each atom has before assigning Formal Charges
C = 4 valence e-, N = v valence e-, Due south = 6 valence east-, also add an extra electron for the (-ane) charge. The full of valence electrons is 16.

iv. Find the most ideal resonance structure. (Note: It is the one with the to the lowest degree formal charges that adds upward to zero or to the molecule'south overall accuse.)
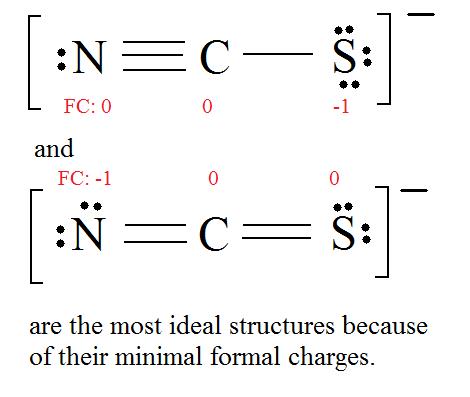
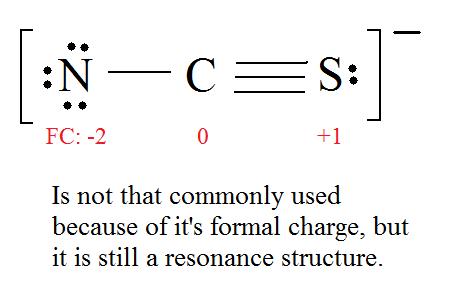
5. Now nosotros have to look at electronegativity for the "Correct" Lewis construction.
The most electronegative cantlet usually has the negative formal charge, while the least electronegative cantlet usually has the positive formal charges.
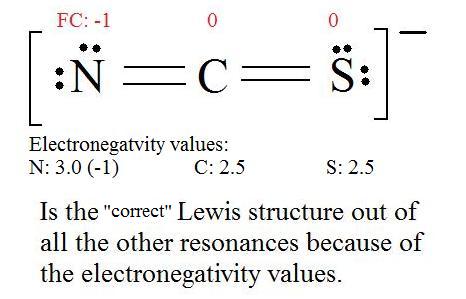
It is useful to combine the resonance structures into a unmarried construction called the Resonance Hybrid that describes the bonding of the molecule. The general approach is described below:
- Draw the Lewis Structure & Resonance for the molecule (using solid lines for bonds).
- Where there can be a double or triple bail, draw a dotted line (-----) for the bail.
- Depict only the lone pairs found in all resonance structures, practise not include the lone pairs that are not on all of the resonance structures.
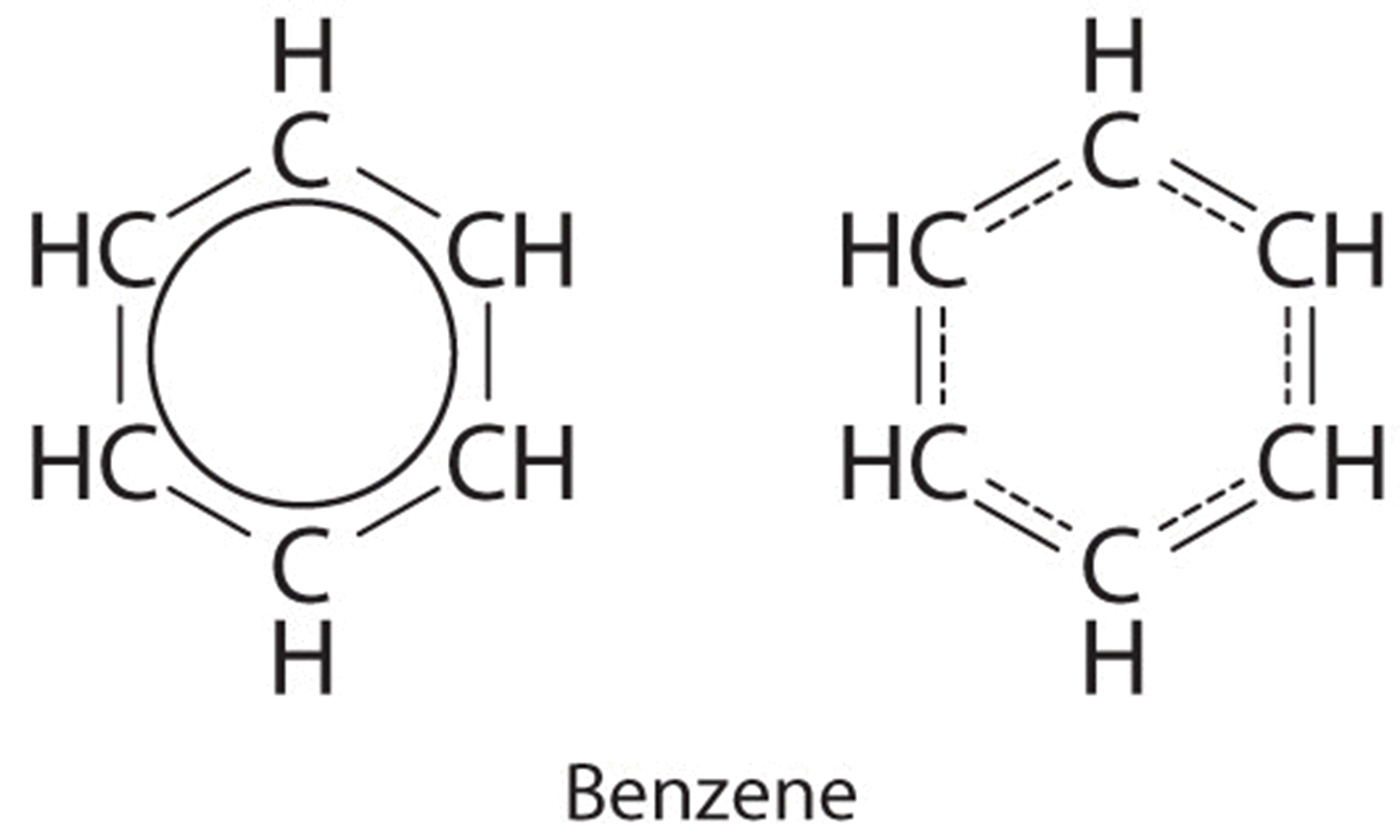
Example \(\PageIndex{four}\): Benzene
Benzene is a common organic solvent that was previously used in gasoline; it is no longer used for this purpose, however, considering information technology is now known to be a carcinogen. The benzene molecule (\(\ce{C6H6}\)) consists of a regular hexagon of carbon atoms, each of which is too bonded to a hydrogen atom. Use resonance structures to depict the bonding in benzene.
Given: molecular formula and molecular geometry
Asked for: resonance structures
Strategy:
- Draw a structure for benzene illustrating the bonded atoms. Then calculate the number of valence electrons used in this drawing.
- Subtract this number from the total number of valence electrons in benzene and then locate the remaining electrons such that each cantlet in the structure reaches an octet.
- Draw the resonance structures for benzene.
Solution:
A Each hydrogen atom contributes ane valence electron, and each carbon atom contributes 4 valence electrons, for a total of (6 × 1) + (vi × iv) = 30 valence electrons. If nosotros place a single bonding electron pair between each pair of carbon atoms and between each carbon and a hydrogen cantlet, we obtain the following:
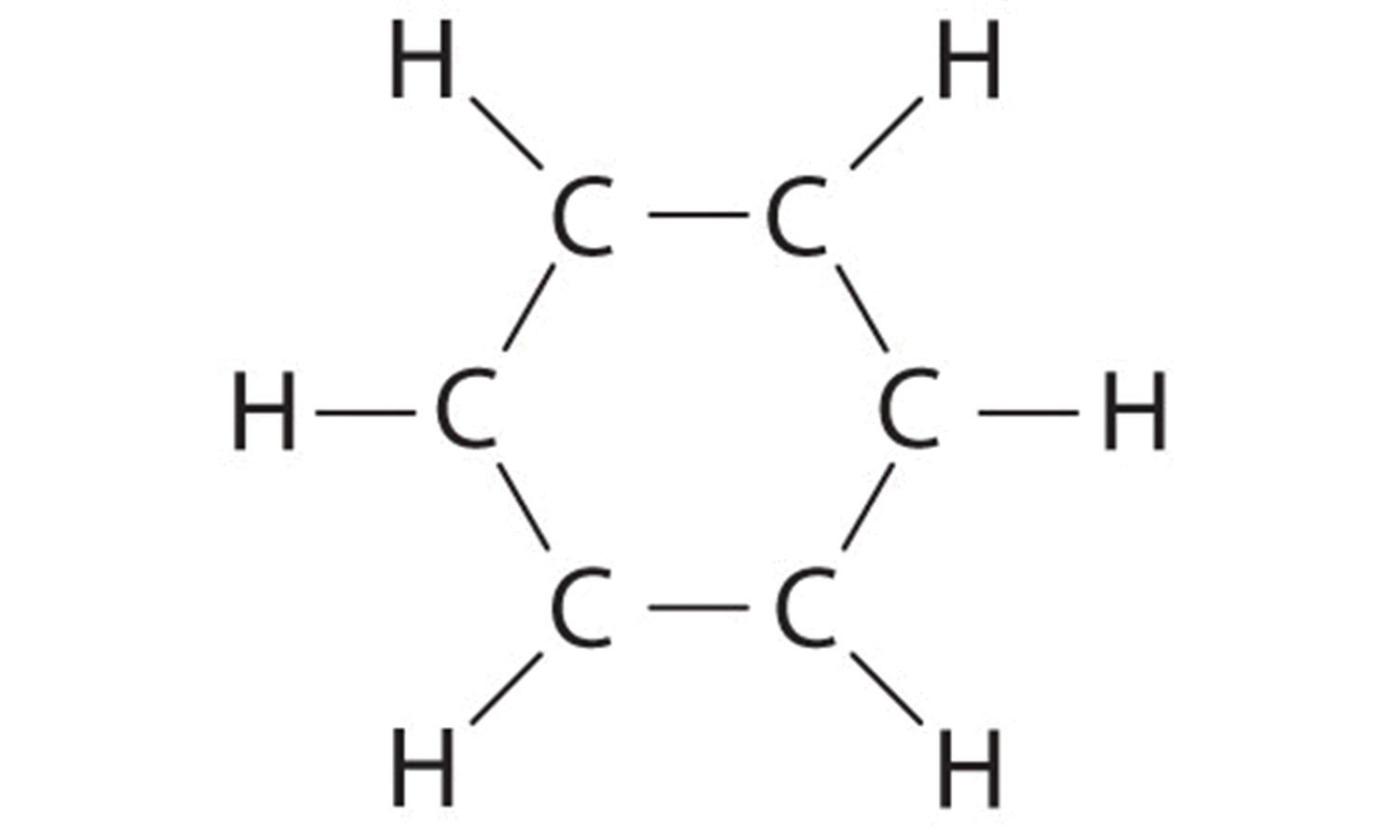
Each carbon atom in this construction has merely 6 electrons and has a formal accuse of +one, but we have used only 24 of the xxx valence electrons.
B If the vi remaining electrons are uniformly distributed pairwise on alternate carbon atoms, we obtain the following:
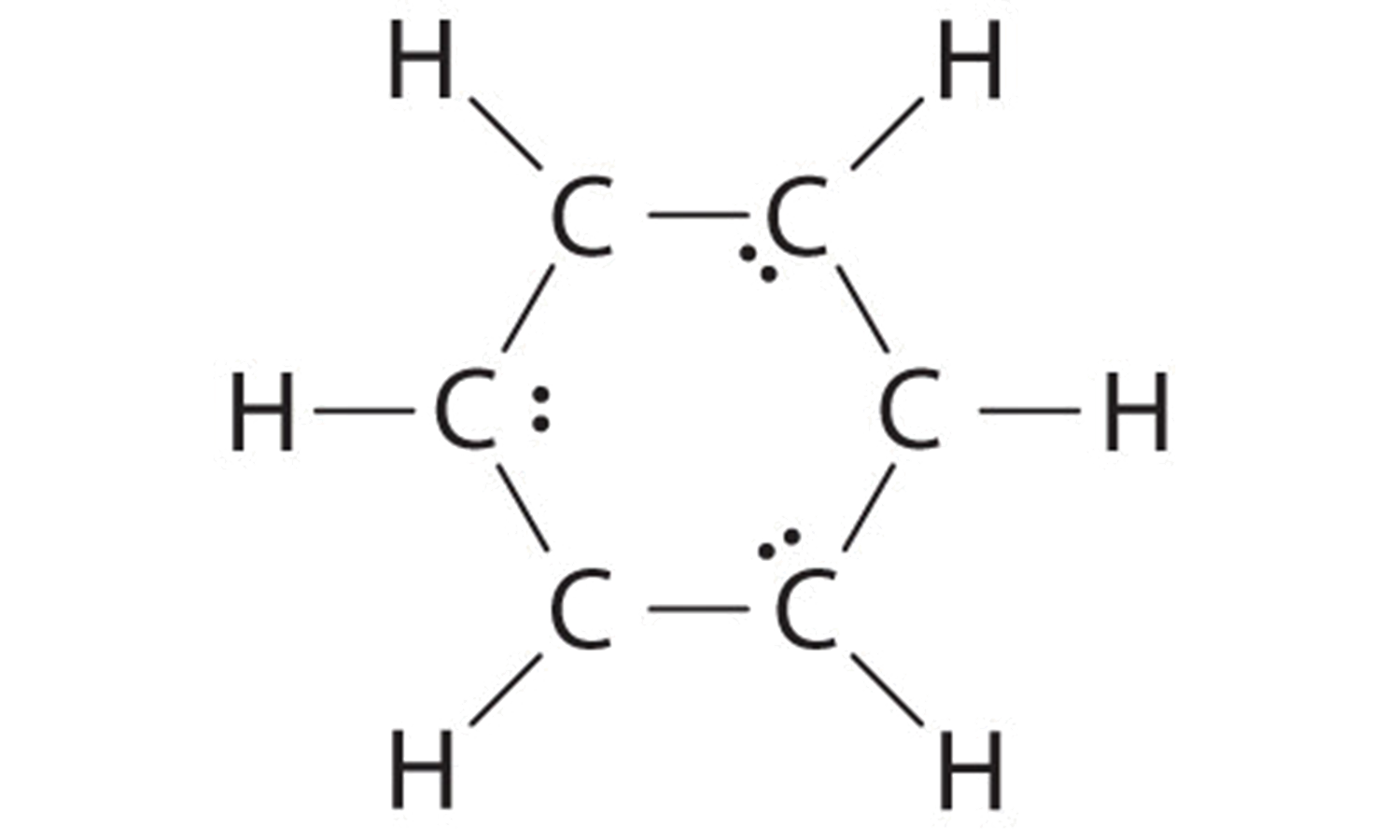
Three carbon atoms now have an octet configuration and a formal charge of −1, while iii carbon atoms have only 6 electrons and a formal accuse of +1. We tin can convert each lone pair to a bonding electron pair, which gives each atom an octet of electrons and a formal accuse of 0, past making three C=C double bonds.
C There are, however, two ways to practise this:
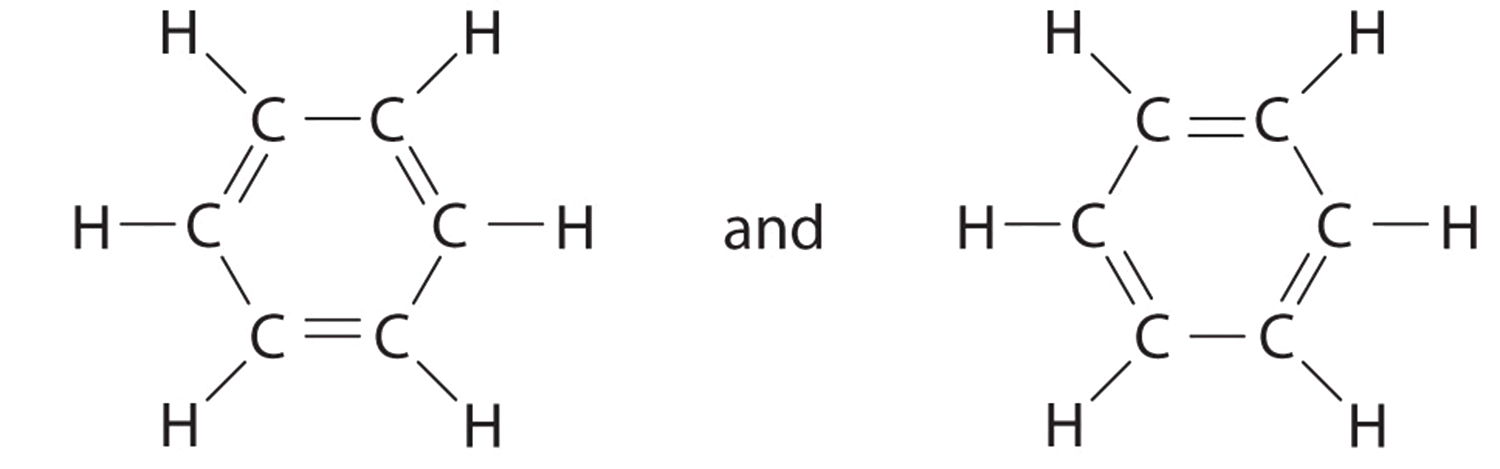
Each structure has alternating double and single bonds, but experimentation shows that each carbon–carbon bail in benzene is identical, with bond lengths (139.nine pm) intermediate between those typically institute for a C–C single bond (154 pm) and a C=C double bail (134 pm). Nosotros tin describe the bonding in benzene using the two resonance structures, but the bodily electronic structure is an average of the two. The beingness of multiple resonance structures for aromatic hydrocarbons like benzene is frequently indicated by cartoon either a circumvolve or dashed lines inside the hexagon:
Example \(\PageIndex{5}\): Nitrate Ion
Draw the possible resonance structures for the Nitrate ion \(\ce{NO_3^{-}}\).
Solution
one. Count upward the valence electrons: (i*five) + (3*six) + i(ion) = 24 electrons
2. Draw the bond connectivities:
3. Add octet electrons to the atoms bonded to the centre atom:
4. Place whatever leftover electrons (24-24 = 0) on the center cantlet:
5. Does the central atom have an octet?
- NO, it has vi electrons
- Add together a multiple bond (first try a double bail) to see if the central atom can achieve an octet:
6. Does the fundamental atom have an octet?
- YES
- Are in that location possible resonance structures? YES
Note: We would wait that the bail lengths in the \(\ce{NO_3^{-}}\) ion to exist somewhat shorter than a single bond.
References
- Petrucci, Ralph H., et al. General Chemistry: Principles and Modernistic Applications. New Jersey: Pearson Prentice Hall, 2007.
- Ahmad, Wan-Yaacob and Zakaria, Mat B. "Cartoon Lewis Structures from Lewis Symbols: A Straight Electron Pairing Approach." Journal of Chemical Education: Journal 77.3.
Problems
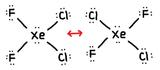
- Truthful or Faux, The film below is a resonance structure?
- Draw the Lewis Dot Structure for SO 4 2 - and all possible resonance structures. Which of the following resonance structure is not favored among the Lewis Structures? Explicate why. Assign Formal Charges.
- Draw the Lewis Dot Construction for CH3COO - and all possible resonance structures. Assign Formal Charges. Choose the most favorable Lewis Construction.
- Draw the Lewis Dot Structure for H PO 3 ii - and all possible resonance structures. Assign Formal Charges.
- Draw the Lewis Dot Structure for CHO2 one - and all possible resonance structures. Assign Formal Charges.
- Draw the Resonance Hybrid Structure for P O four three - .
- Draw the Resonance Hybrid Construction for North O three - .
Problems #2
Answers
1. False, considering the electrons were non moved around, but the atoms (this violates the Resonance Structure Rules).
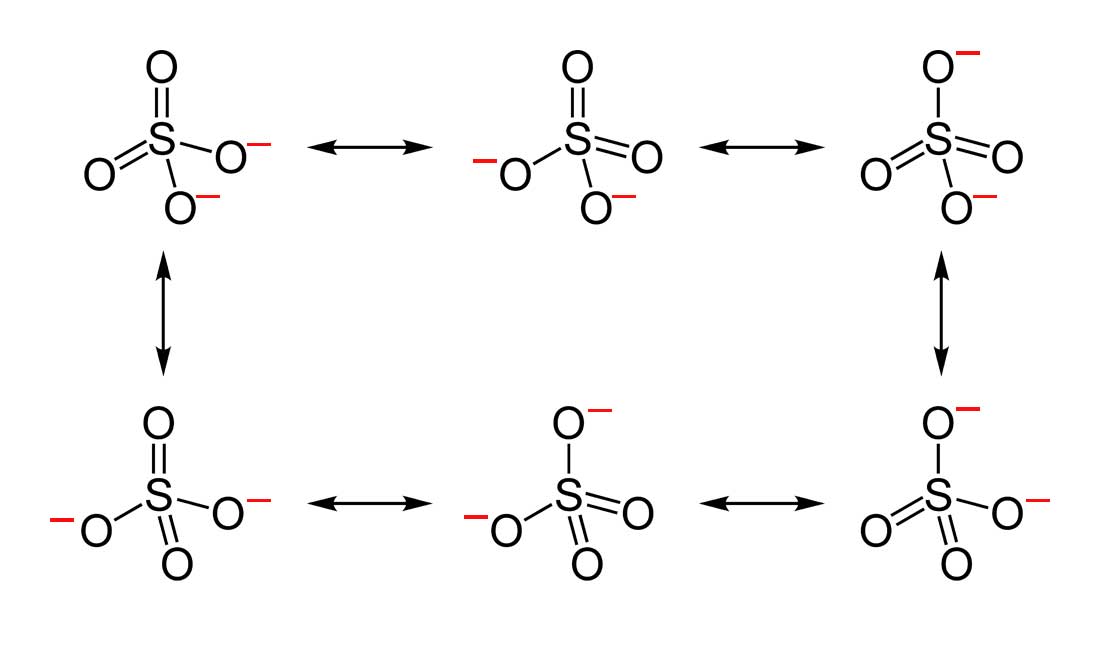
two. Below are the all Lewis dot structure with formal charges (in red) for Sulfate (SO4 two - ). There isn't a virtually favorable resonance of the Sulfate ion because they are all identical in charge and in that location is no change in Electronegativity between the Oxygen atoms.
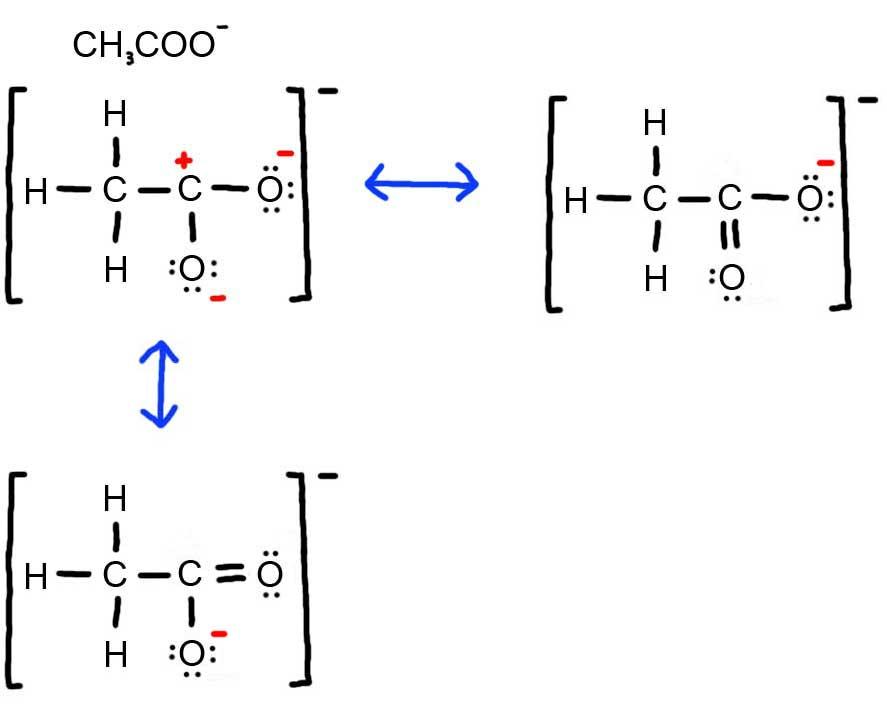
iii. Below is the resonance for CH3COO -, formal charges are displayed in red. The Lewis Structure with the most formal charges is not desirable, because we want the Lewis Structure with the least formal charge.
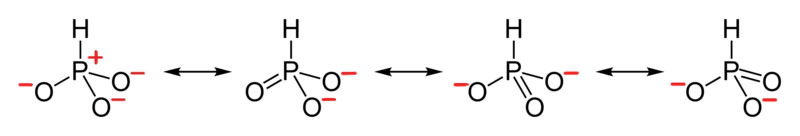
iv. The resonance for HPO3 2 - , and the formal charges (in red).
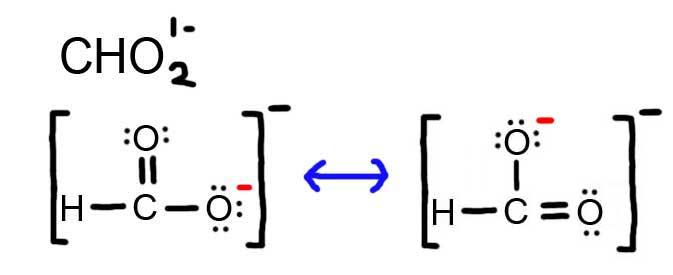
5. The resonance for CHO2 1 - , and the formal charges (in cerise).
half-dozen. The resonance hybrid for PO four 3 - , hybrid bonds are in red.
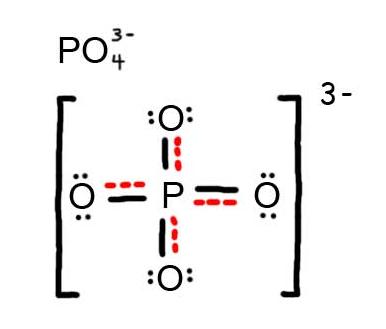
7. The resonance hybrid for NO iii - , hybrid bonds are in red.
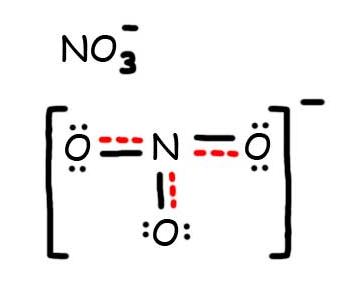
Problems #2
Contributors and Attributions
- Sharon Wei (UCD), Liza Chu (UCD)
Source: https://chem.libretexts.org/Bookshelves/Physical_and_Theoretical_Chemistry_Textbook_Maps/Supplemental_Modules_%28Physical_and_Theoretical_Chemistry%29/Chemical_Bonding/Valence_Bond_Theory/Resonance
Posted by: barnescamonwarld1947.blogspot.com


0 Response to "draw the lewis structure for so42-. how many equivalent resonance structures can be drawn?"
Post a Comment